Papsdorf Lab
Research Publications Group Members BiographyLipid homeostasis in ageing and longevity
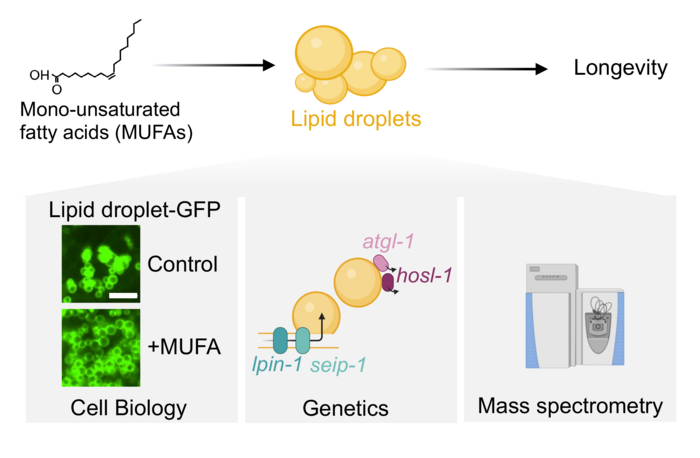
Ageing is characterised by the gradual decline of tissue function. This decline also increases susceptibility to multiple diseases, including cardiovascular diseases, neurodegenerative disorders and cancer. Age progression can be slowed by interventions such as diet. One key component of the diet are lipids, which are essential for cellular homeostasis as they serve as energy reservoirs and constitute the building blocks of membranes. In the context of ageing, specific lipids are potent modulators of lifespan. For example, diets rich in monounsaturated fatty acids (MUFAs), such as the Mediterranean Diet, improve blood profiles associated with diabetes and correlate with longevity in humans. Specific MUFAs (e.g. oleic acid and palmitoleic acid) also causally extend lifespan in C. elegans. This suggests that lipids are a powerful target to analyse and manipulate in the context of ageing.
The overarching goal of my lab is to decipher how specific lipids drive cellular changes that induce longevity. We focus on the cellular organelles controlling these processes. To study the connection between lipids and ageing, we use the nematode C. elegans. The nematode worm is uniquely positioned because many of the pathways and organelles involved in lipid processing are conserved with mammals and its short lifespan allows us to perform ageing studies in a laboratory setting. We use a combination of cell biology and mass spectrometry coupled with genetics and screening techniques to dissect the functions of specific lipids and the organelles that process them for ageing and longevity.
Lipid droplet impact on tissue homeostasis during ageing and longevity
Lipid droplets are conserved organelles that are critical for energy homeostasis. For a long time, lipid droplets were considered inert storage organelles, however recent studies discovered that they are highly dynamic, with specific cellular machinery regulating different aspects of their biology. We recently showed that an increase in lipid droplets is critical for longevity upon treatment with MUFAs. We now focus on unravelling the mechanism of how lipid droplets shape tissue and cellular homeostasis to drive longevity. To do this, we use a combination of tissue-specific and dietary manipulations, mass spectrometry and cell biology to understand how lipid droplets drive longevity in a tissue-specific manner.
Organelle contact changes during ageing and longevity
Organelles rarely act in isolation, and they are in frequent contact with each other to exchange lipids, proteins or metabolites. However, despite their critical role in numerous biological processes, it is unknown how ageing affects the organelle interaction landscape and if such interactions can be leveraged to prolong lifespan. We are interested in identifying changes in the organelle interaction landscape with ageing and how they differ in long-lived individuals, as well as in exploring whether targeting organelle interactions could extend lifespan. To do this, we will use a combination of genome-wide screening tools, fluorescence microscopy, electron microscopy and protein engineering to find key organelle interactions for lifespan.
Regulation and functional importance of ether lipids during ageing
Ether lipids are a specific type of membrane lipid that are biomarkers of ageing and often dysregulated in disease. These abundant lipids can constitute up to 20% of all membrane lipids. However, the precise organelle membrane hosting these lipids and their impact on lifespan remains largely unknown. We are interested in determining the cellular localisation of ether lipids and unravelling their functional importance in organelles and lifespan regulation. To address these important questions, my lab uses a combination of organelle isolations, biochemistry, mass spectrometry, genetic manipulations and lifespan studies.
For more information, please visit the Papsdorf Lab website.
Recruiting
We are always interested in motivated researchers from all levels looking to join our team. If you find our research interesting and would like to apply, please send us an email.